SEE® Trabecular Acetabular Cup System
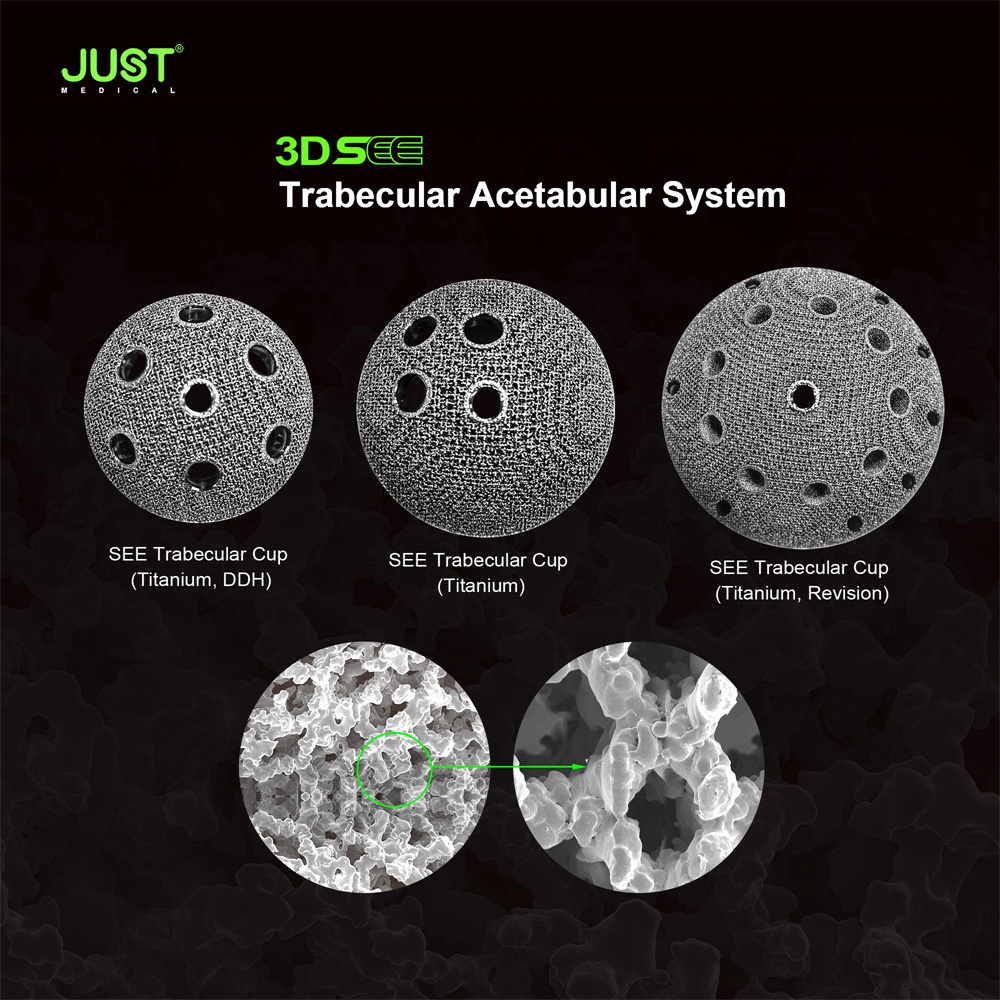
SEE Trabecular Acetabular Cup System
Product Features:
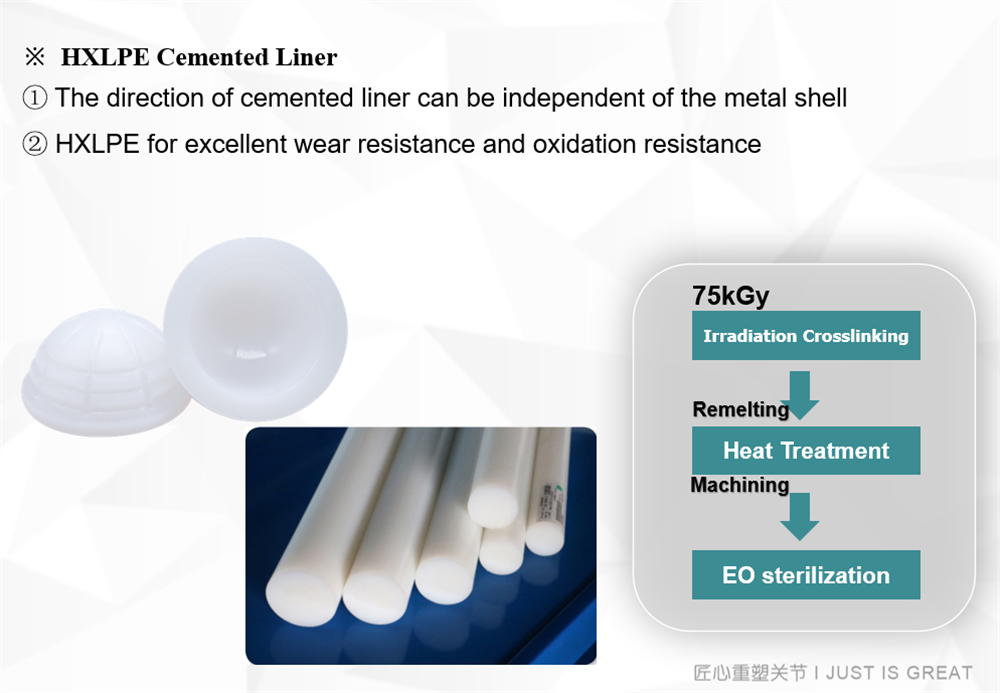
Multiple liner options
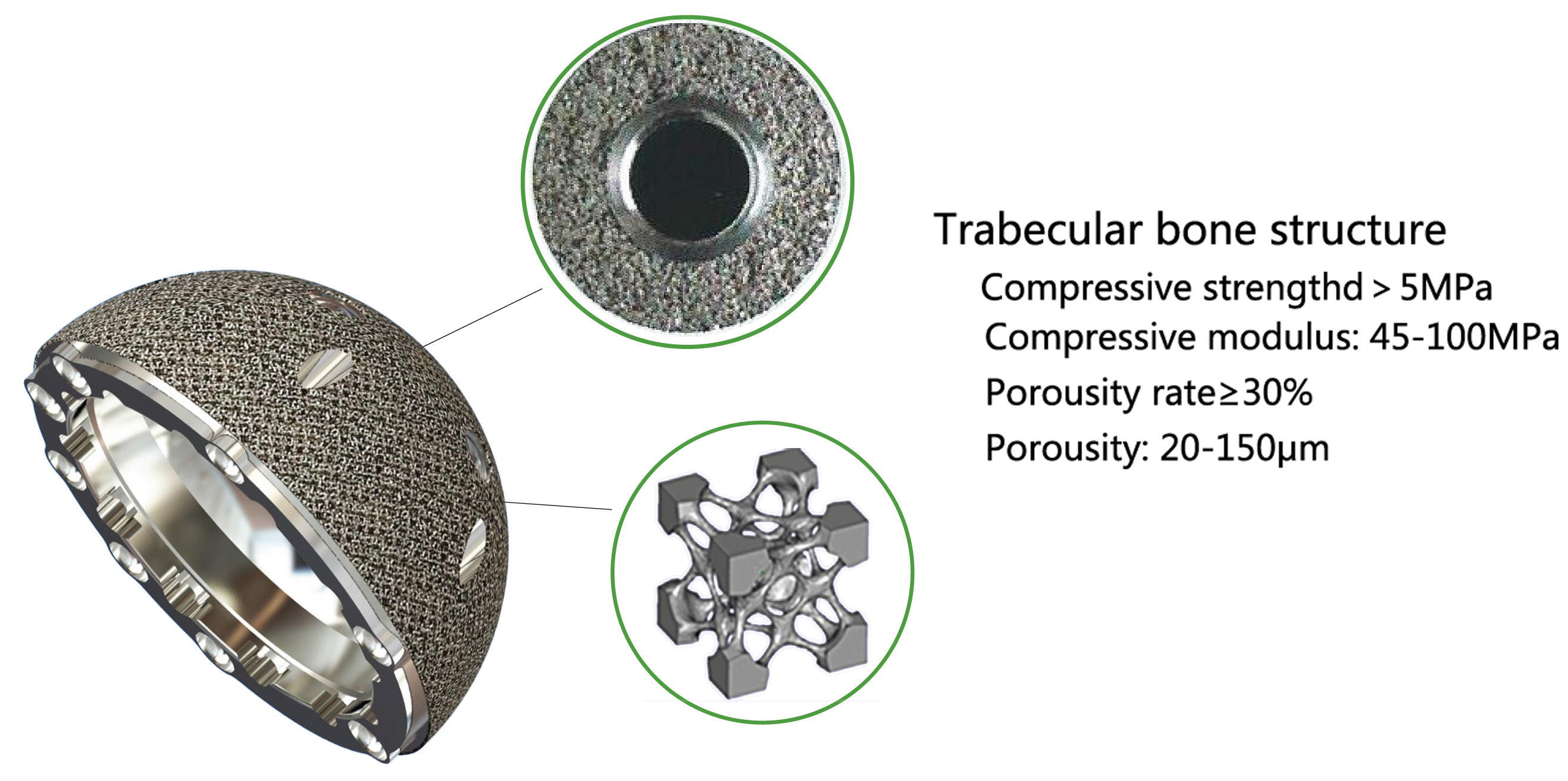
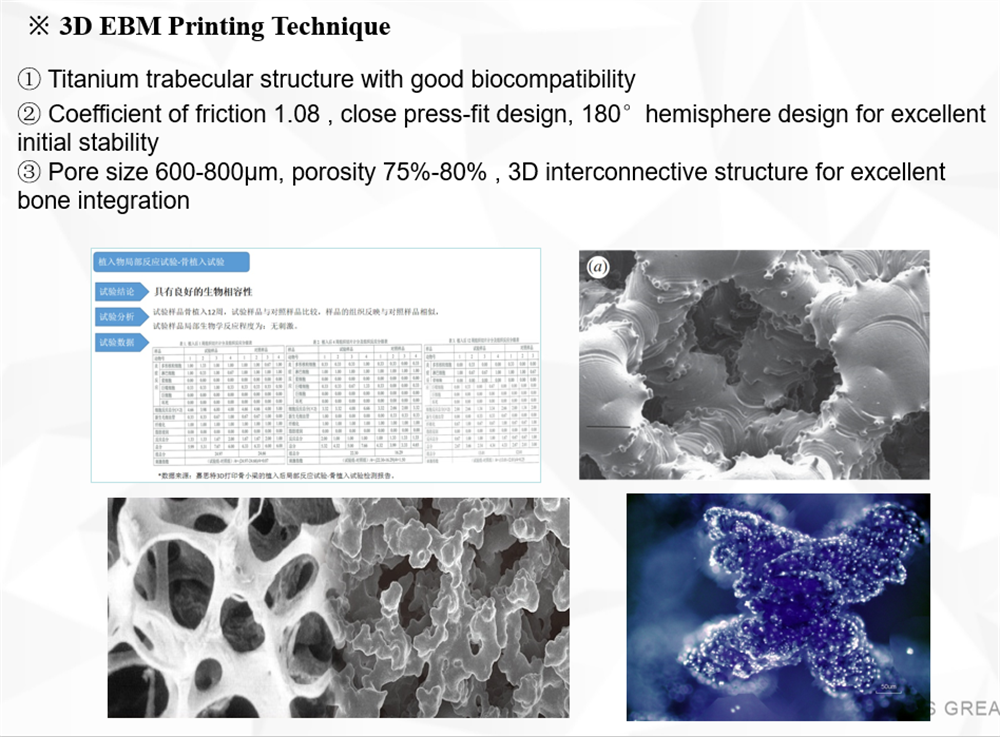
Electron beam melting technology reduces structural adhesion interface, reduces wear risk and facilitates bone integration;
80% porosity, 600-800μm three-dimensional joint micropores, which are beneficial to bone tissue deposition and achieve three-dimensional binding;
the elastic modulus of the cancellous bone, avoiding stress shielding and bone resorption;
retain the HARMONY cup of multi-liner design;
Material: Ti6Al4V
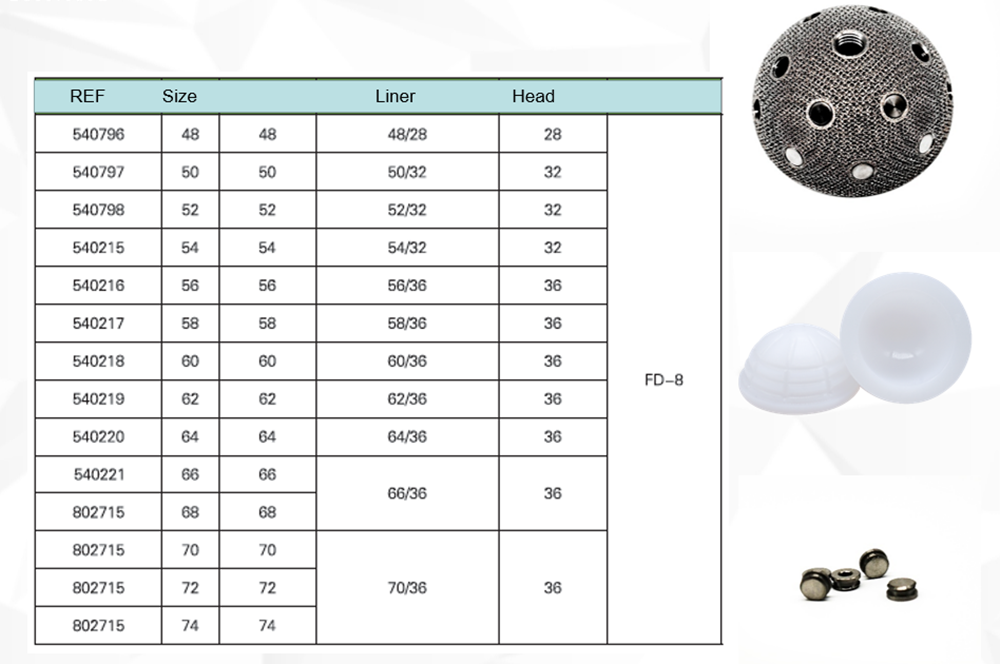
Specification: DDH: 46MM; STANDARD: 48-66MM; REVISION: 54-66MM


Parameters
If You Have Any Questions, Click Here To Submit The Form
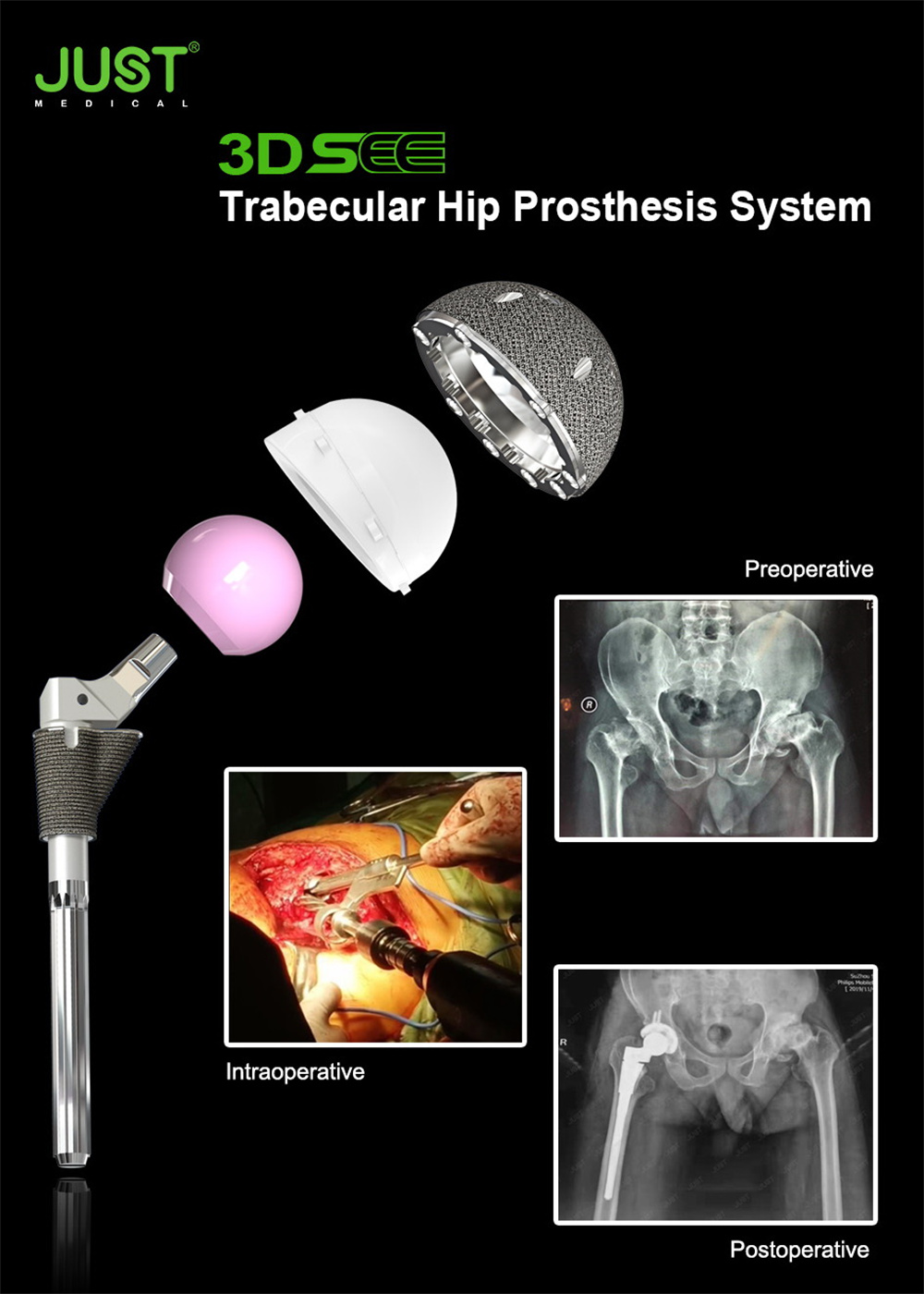
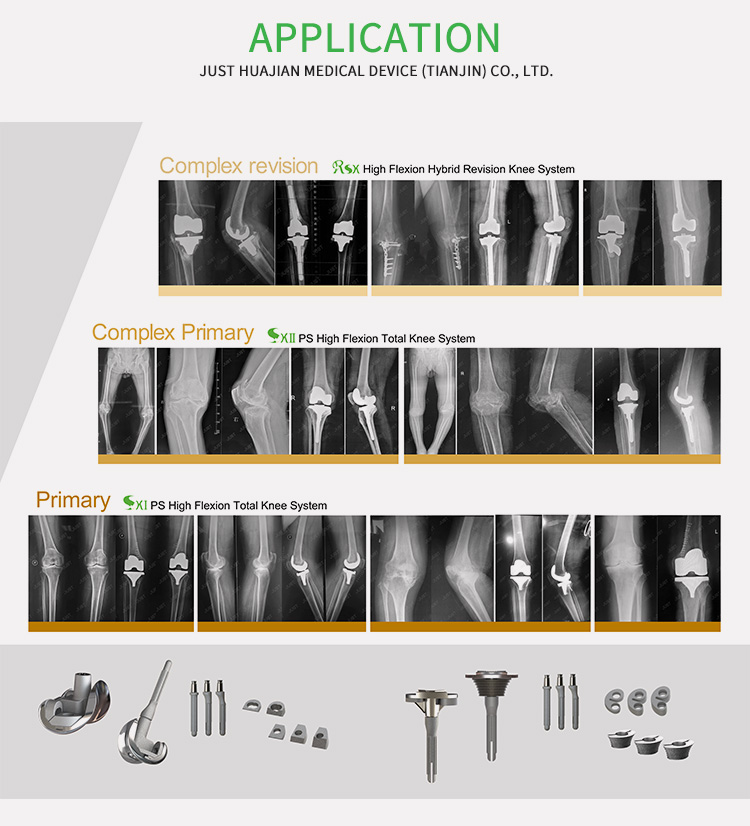
Real Case
Product Training Support

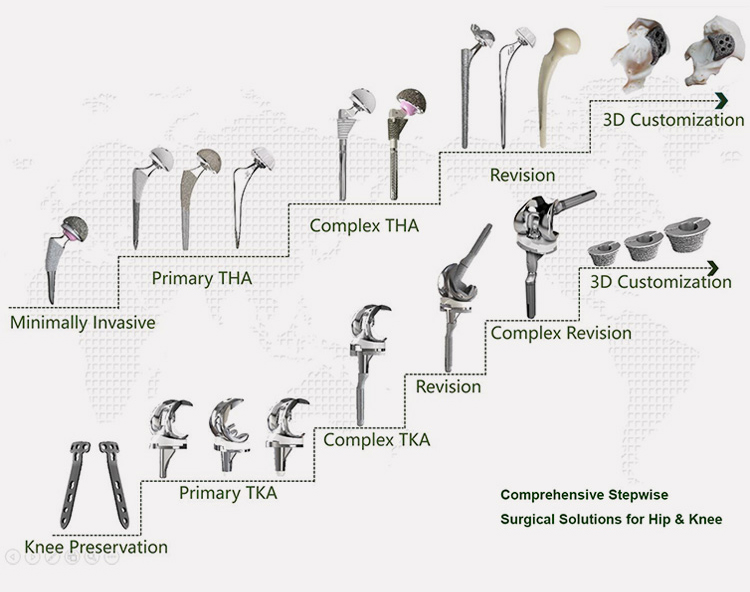
Whole Product line
Production Line

5 Million Cycles Wear Test
for TKR and THR System in ENDOLAB,Germany

10 Million Cycles Fatigue Test
for Femoral Stem (Neck) and Tibial Tray

Imported Raw Material

Quality Assurance

Services
Prompt response to enquiry in 24 hours.
Competitive prices and no MOQ requirement to help customers test the quality and market.
High quality with ISO, CE certified.
Proficient delivery and export handling.
Surgical technique training and workshop in China and sending surgeons to demonstrate and support surgery for distributors.
Sponsorship in advertisement and expo.
Training course for technicians of distributors CME meetings

Annual Orthopedic Meeting

Business Partners

-
JUST Medical Knee Joint Prosthesis System - SKII PS & SKII PLUS Se...
Dec. 21, 2023
-
Case sharing | 3D Printing Empowering Digital Orthopedics
Nov. 01, 2023
-
Expert Consensus: Choice for UKA Patients
Nov. 01, 2023
-
How can dual mobility total hip arthroplasty effectively reduce the dislocation rate?
Nov. 01, 2023